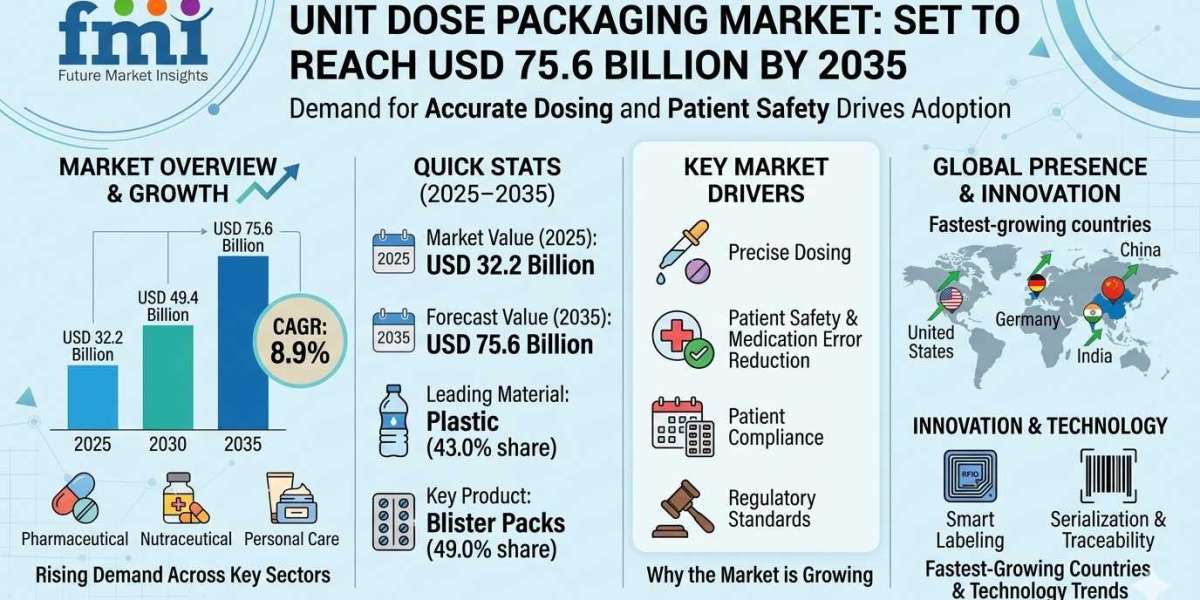
The global Unit Dose Packaging Market is projected to expand significantly, rising from an estimated USD 32.2 billion in 2025 to USD 75.6 billion by 2035, reflecting a robust compound annual growth rate (CAGR) of 8.9%. This growth is underpinned by increasing demand for precise dosing solutions across pharmaceutical, nutraceutical, and personal care products. Rising awareness regarding medication safety, combined with stringent regulatory standards, is accelerating adoption across both hospital and retail pharmacy channels.
Between 2025 and 2030, the market is expected to grow steadily from USD 32.2 billion to USD 49.4 billion. Enhanced patient compliance initiatives, coupled with adoption of blister packs, strip packs, and pre-measured sachets, are shaping industry transformation. The integration of smart labeling, serialization, and traceability technologies further supports adoption, positioning unit dose packaging as a critical element in modern healthcare supply chains and sustainable packaging innovation strategies.
Quick Stats: Unit Dose Packaging Market (2025–2035)
- Market Value (2025): USD 32.2 Billion
• Forecast Value (2035): USD 75.6 Billion
• CAGR: 8.9%
• Leading Segment: Plastic (43.0% share)
• Key Product Segment: Blister Packs (49.0% share)
• Fastest-Growing Countries: China, India, Germany, France, United States
• Key Growth Driver: Demand for precise and safe dosing solutions
Structural Growth Driver: Patient Safety and Regulatory Compliance
The growth of the unit dose packaging market is strongly influenced by regulatory mandates and safety-driven adoption. Healthcare regulations globally emphasize patient safety, accurate dosing, and tamper-evident packaging, prompting manufacturers to innovate and comply with industry standards. Hospitals and pharmacies increasingly adopt unit dose formats to reduce medication errors and enhance adherence, particularly for high-risk drugs and chronic therapy management.
Technological and environmental pressures further shape the market, as manufacturers implement recyclable materials, compact formats, and sustainable production processes. Compliance with international standards is critical, with companies seeking certifications and adherence to good manufacturing practices (GMP).
- Recycling regulations
• Certification requirements
• Material sustainability guidelines
• Manufacturing standards
From Compliance to Industry Transformation
Pharmaceutical and personal care companies are transforming operations to align with evolving regulatory expectations. Automation, digital compliance tools, and smart labeling technologies are being integrated into packaging workflows to enhance precision and efficiency. The market is seeing widespread adoption of single-dose solutions, driven by patient-centric care models and hospital safety protocols.
Companies prioritize optimizing supply chains while balancing cost and compliance pressures. Integration of pre-filled syringes, tamper-evident packs, and portion-controlled sachets is enhancing operational flexibility and consumer convenience.
- Sustainability performance
• Cost efficiency
• Recyclability
• Operational compatibility
Technology Transformation: Smart and Sustainable Packaging Innovation
Technological advancements in materials and manufacturing processes are central to the unit dose packaging market. The adoption of advanced plastics, bioplastics, and high-barrier films ensures dose integrity and shelf-life stability. Automation and robotics in production lines increase throughput while reducing errors, while smart labels and serialization enable real-time traceability.
- Advanced material development
• Automation integration
• Lightweight design
• Sustainable material engineering
• Manufacturing efficiency improvements
Segment Highlights
By Material Type
• Plastic (43.0% share): Flexible, lightweight, and compatible with automated production lines
• Aluminum (22.0% share): Barrier properties suitable for sensitive formulations
• Paper Paperboard (18.0% share): Eco-friendly option for dry oral medications
• Glass (17.0% share): Preferred for injectables and high-purity liquids
By Packaging Type
• Blister Packs (49.0% share): Tamper-evident, child-resistant, widely used for oral drugs
• Sachets Pouches (22.0% share): Single-dose liquid formulations for nutraceuticals
• Ampoules Vials (15.0% share): Sterile packaging for injectables and biologics
• Pre-Filled Syringes (10.0% share): Increasing adoption for outpatient injections
• Others (4.0% share): Niche formats for specialty applications
Regional Outlook: Emerging Economies Drive Adoption
Unit dose packaging is witnessing accelerated growth across Asia-Pacific and Europe, with developing economies adopting scalable, compliant packaging solutions. Regulatory support, rising healthcare expenditure, and expanding pharmaceutical production in emerging markets are key drivers of regional expansion.
- India (11.1% CAGR): Growth driven by affordable healthcare programs and generic drug adoption
• China (12.0% CAGR): Expansion fueled by pharmaceutical production and regulatory enforcement
• Brazil (6.7% CAGR): Rising demand in hospitals and retail pharmacies
• Germany (10.2% CAGR): Focus on high-quality, sustainable packaging formats
• United States (7.6% CAGR): Mature market with strong adoption of digital and smart compliance solutions
Risk Landscape: Market Constraints and Challenges
While growth prospects remain strong, the unit dose packaging market faces challenges that could constrain expansion. High production costs, complex machinery requirements, and strict regulatory frameworks limit small-scale adoption. Supply chain vulnerabilities and raw material volatility pose additional risks.
- Raw material price volatility
• Supply chain disruptions
• Infrastructure gaps
• Regulatory complexity
• High manufacturing costs
Competitive Landscape: Key Market Players
The competitive landscape is characterized by consolidation and innovation, with leading players leveraging technology, sustainable solutions, and global manufacturing presence to strengthen market position. Companies focus on precision dosing, smart packaging, and patient-centric solutions to gain competitive advantage.
- Amcor
• West Pharmaceutical Services
• Catalent
• Gerresheimer
• AptarGroup
Other notable companies include Adelphi Healthcare Packaging, Berry Global, Borosil, Constantia Flexibles, Corning, DWK Life Sciences, Glenroy, Lameplast, Nipro, Schott, SGD Pharma, Shiotani Glass, Stevanato Group, Unit Pack Company, and Valmatic.
Outlook: Future of the Unit Dose Packaging Market
The unit dose packaging market is poised for sustained growth over the next decade, driven by regulatory pressure, technology adoption, and patient safety imperatives. Expansion in emerging markets, integration of smart and connected packaging solutions, and development of sustainable formats will continue to shape the competitive and operational landscape.
- Technology advancement
• Sustainability initiatives
• Manufacturing expansion
• Supply chain innovation
visit the official report page at: https://www.futuremarketinsights.com/reports/unit-dose-packaging-market